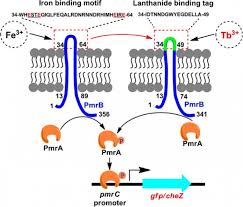
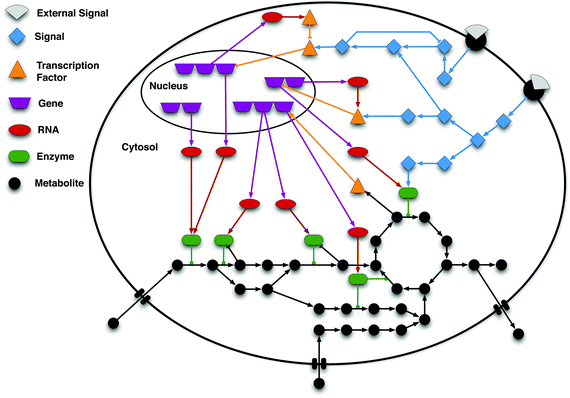
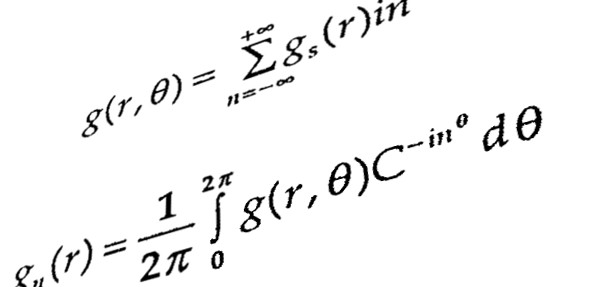Biochemical processes are simply complex, and their apparent feature does not easily allow us to investigate what exactly system means. Moreover, most of the biochemical processes obey nonlinear reaction kinetics. That is, amount of reactant (Protein/RNA/DNA/) is not directly proportional to its product. This leads to further increase in complexity level of molecular mechanism. And create biological noises such as randomization (stochasticity) of biomolecules, perturbation in cell signaling, difficulty in quantification of cell products and even unexpected response of the entire system. Here comes development and utilization of a mathematical model takes multiple factors/parameters into consideration and provides researcher with a visual understanding how complex biological system functions and responds to external (hormone/drug/cation/anion) and internal signals (protein/enzyme/cation/anion) or adverse environmental condition such as deficiency of Fe2+ ion during formation of Vitamin-D. Basically, mathematical modeling receives a broad domain of cellular processes such as metabolic regulation, gene-gene interaction, and gene-protein interaction. This has made a bridge between experimental and expected outcome. In case of discrepancies, between the two, parameter taken into consideration need to be refined. The general approach of the modeling give us the following benefit:
- Discrepancies between mathematical model and actual experimental result point to components that still are missing from hypothetically developed model, and therefore one can develop a more comprehensive scenario of systems behavior. On the hand, a well developed model assist in designing and clarifying the additional issues in ongoing experiment.
- With the help of a mathematical model researcher can modify experimental parameter (e.g., by introducing modified protein associated with Mg2+ uptake into cell) and run the computer simulations.
- Most importantly, mathematical models are not limited to environmental/experimental constraints. They may be quickly changed for multiple conditions/parameters and most suitable simulation can be assessed for developing a reliable experimental design.
- A mathematical model may help to investigate sub-system that sometimes regulate special biochemical process, though all biological reactions cannot be treated same, which necessarily provides substantial information about large systems behaviour.
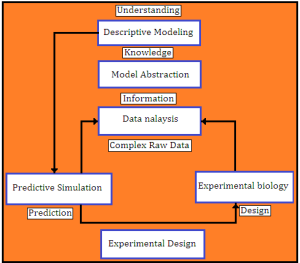
By doing multiple simulations and changing parameter values, we are able to represent the real biochemical/molecular phenomena which seem to be difficult-to-treat.
→ Continue to Introduction to Mathematical Modelling – Part 2