Systems pharmacology is an emerging area in the field of medicinal chemistry and pharmacology which utilizes systems network to understand drug action at the organ and organism level. It applies the computational and experimental systems biology approaches to pharmacology, which includes network analyzes at multiple biological organization levels facilitating the understanding of both therapeutic and adverse effects of the drugs. Nearly a decade ago, the term systems pharmacology was used to define the drug action in a specific organ system such as reproductive pharmacology [1], but to date, it has been expanded to different organ and organism levels [2].
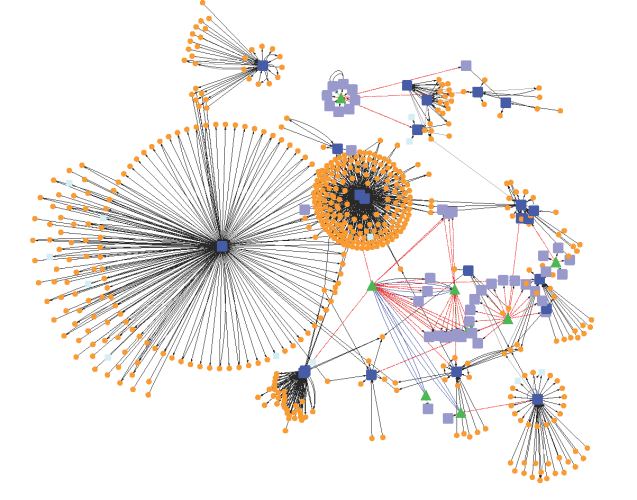
Network approaches are useful in organizing large biological datasets and obtaining useful information. A network represents the relationship between the nodes, which represents proteins [3], genes [4], drugs [5], and diseases [6,7]. The nodes are connected through the edges which represent protein-protein interaction [3], drug-drug interaction [8], and so on (in the context of drug action).
The computation tools provide network analysis methods for identifying potential drug targets, which can later be used to develop drugs. Network analysis methods provide the number of proteins getting affected by targeting a particular protein in the network and help to identify whether the protein participates in motif or not [9]. These methods can be used to predict the positive and negative effects of the potential drug. For example, a protein with high connectivity may cause lethal effects, and the disease genes with low connectivity are lethal but may lead to disease phenotype [10]. The positive and negative effects of the potential drugs can be studied by analyzing the proximity of a protein to another protein which is involved in the long QT interval (a measure of the time between the start of the Q wave and the end of the T wave in the heart’s electrical cycle) event. A network algorithm applying MFPT (mean first passage time) can be used to identify the protein that may lead to long QT interval [11]. The nearest neighbor method can be used which assesses the distance by measuring the shortest path between the protein of interest to any other known proteins leading to long QT event.
Another advantage of using systems network in the context of the targets involved in therapeutic and adverse actions. Network systems can identify physiologically relevant targets and determine the other nodes with which these targets show their action. Similarly, there are some databases available which store information related to all drugs and their effects such as DrugBank [8]. A database, FDA Adverse Events Reporting System (AERS) is a publicly available database which stores drug-induced adverse events in the patients using one or more drugs. It helps in building organ-level and organism-level networks through which the concurrence of therapeutics and related adverse phenotype can be identified.
The network analysis can identify the potential drug targets at the macromolecular level, they need to be filtered by structural criteria to determine their ability at the atomic-level. The development of new drugs requires deep insight into the structures of the targets and the drugs which help to study the atomic-level interactions. The binding pockets of the target are analyzed in order to determine the best fit pocket for the drug, in some cases when the binding pockets do not accept the drug, then the structure of the drug is modified. This is a well-studied area in pharmacology which uses computer aided drug designing (CADD). This helps in either activation or inhibition of the target depending on the need. Further, the ADMET (absorption, distribution, metabolism, excretion, and toxicity) properties can be predicted.
The systems pharmacology has an enormous impact on drug development and drug usage which can provide means to develop drugs for the diseases such as type 2 diabetes, cancer, other metabolic and immune disorders which are not susceptible to simple treatment approaches involving the use of antibiotics.
References
1. Brunton, L., Lazo, J., & Parker, K. G. (2005). Gilman’s The Pharmacological Basis of Therapeutics. 11th.
2. Zhao, S., & Iyengar, R. (2012). Systems pharmacology: network analysis to identify multiscale mechanisms of drug action. Annual review of pharmacology and toxicology, 52, 505-521.
3. Stark, C., Breitkreutz, B. J., Chatr-Aryamontri, A., Boucher, L., Oughtred, R., Livstone, M. S., … & Reguly, T. (2011). The BioGRID interaction database: 2011 update. Nucleic acids research, 39(suppl 1), D698-D704.
4. Davidson, J. R., Zhang, W., Connor, K. M., Ji, J., Jobson, K., Lecrubier, Y., … & Stein, D. J. (2010). Review: A psychopharmacological treatment algorithm for generalised anxiety disorder (GAD). Journal of Psychopharmacology, 24(1), 3-26.
5. Zhao, S., & Li, S. (2010). Network-based relating pharmacological and genomic spaces for drug target identification. PloS one, 5(7), e11764.
6. Wang, X., Gulbahce, N., & Yu, H. (2011). Network-based methods for human disease gene prediction. Briefings in functional genomics, 10(5), 280-293.
7. Vidal, M., Cusick, M. E., & Barabasi, A. L. (2011). Interactome networks and human disease. Cell, 144(6), 986-998.
8. Wishart, D. S., Knox, C., Guo, A. C., Cheng, D., Shrivastava, S., Tzur, D., … & Hassanali, M. (2008). DrugBank: a knowledgebase for drugs, drug actions and drug targets. Nucleic acids research, 36(suppl 1), D901-D906.
9. Ma’ayan, A., Jenkins, S. L., Neves, S., Hasseldine, A., Grace, E., Dubin-Thaler, B., … & Kershenbaum, A. (2005). Formation of regulatory patterns during signal propagation in a mammalian cellular network. Science, 309(5737), 1078-1083.
10. Wang, X., Gulbahce, N., & Yu, H. (2011). Network-based methods for human disease gene prediction. Briefings in functional genomics, 10(5), 280-293.
11. Berger, S. I., Ma’ayan, A., & Iyengar, R. (2010). Systems pharmacology of arrhythmias. Science signaling, 3(118), ra30.